My previous blog highlighted the useful cases of iHelp. This blog will examine how to develop content and implement iHelp.
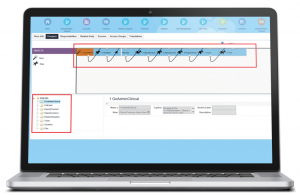
iHelp is developed and administered through the Administration iHelp screen. The Designer view provides a Java-based workflow editor, which is very much like a simple version of Microsoft Visio. You drag the steps onto the grid and connect those using arrows. The steps then appear in the applets below. The Designer view provides an explorer tree to navigate your way around the elements of each iHelp article, and a form applet to enter the content itself.
For example, you can see the step “Name” (which is only seen by developers), as well the “View,” which specifies the target of the view link (if included), and governs which business components are available to select field and button highlights from.
Training one or two users to develop content in iHelp could help the rest of your users become experts in Siebel CTMS, minimizing your need for refresher training, reference materials, and user support. However, if these roles are fulfilled by different people, you can set up the steps in a text or Word document, and then the iHelp administrator can enter these into your system.
To learn more about iHelp and the assistance it can provide with Oracle’s Cloud Siebel CTMS, you can click here or fill out the form below.

