In my previous blog post entitled Using On-demand Data With Dynamic Data Writeback In Spotfire, I discussed using Spotfire as an aid for Clinical Review of data along with supporting writeback capabilities to record the user review actions. As a follow-on discussion, I wanted to expand those capabilities to include Clinical Data Cleaning Review in conjunction with Data Manager Data Cleaning Review.
Before statistical analysis and reporting of clinical data for regulatory agencies can begin, Clinicians and Medical Monitors must review the state of the cleanliness of the data in collaboration with the Data Managers. After the Data Managers have used their tools to establish the cleanliness of the data, the clinical data and cleanliness state information is used by the Clinicians and Medical Monitors to perform a clinical review of the data. Once the cleanliness state is accepted by the Clinician/Medical Monitor, the data is ready for formal reporting.
In discussing a process for reviewing data for Clinician/Medical Monitor Data Cleanliness Review, a number of ways to improve the Clinical Data Review process will be raised, including:
- Using multiple sources – source data (Oracle Data Management Workbench InForm, Oracle Clinical, external lab data, etc.) and derived Value Added Data
- Importing externally managed data as well as internally managed data
- Integrating with internal and external query management tools
- Using Visualization Tools
In my next few blog posts, we will look at various scenarios for reviewing clinical data cleanliness.
Reviewing Clinical Data Cleanliness In Oracle DMW
Today, we’ll take a look at a simple scenario for reviewing clinical data cleanliness, which entails a clinical or medical monitor review of data from Oracle’s Data Management Workbench.
Let me note that the graphic above is a logical data flow representation. Many of the features I will be describing are built into Oracle’s DMW with regard to creating derived data and maintaining data cleanliness state information along with associated data lineage. For this discussion, we will logically separate the Clinical/Medical Monitor data cleanliness review from the data manager data cleanliness data review in DMW.
In the data flow above, the Medical Monitor Data Evaluation process, uses data from DMW to review the data and record the data review state of the data. This should include a Changed Since Last Review usability feature for supporting the data review state for each row of data (new, changed since last review, etc.). For data that has changed since the last review, the changed data should be highlighted and review audit information provided.
This audit information should include when the data was last reviewed and the data values at that point in time for comparison. This enables the Clinician/Medical Monitor to see what data has changed and when. Providing associated query information helps round out the picture of the state of the data for the Clinician/Medical Monitor.
Additionally, a feature of allowing the Clinician/Medical Monitor to create new data queries against questionable data points can act as a communication vehicle with the Data Manager to aid in resolving questionable data.
This data review should include a number of usability features:
- Provide simple data listings with state information (more on that below)
- Use filters and Sorting
- Provide additional derived data (discussed in greater detail below)
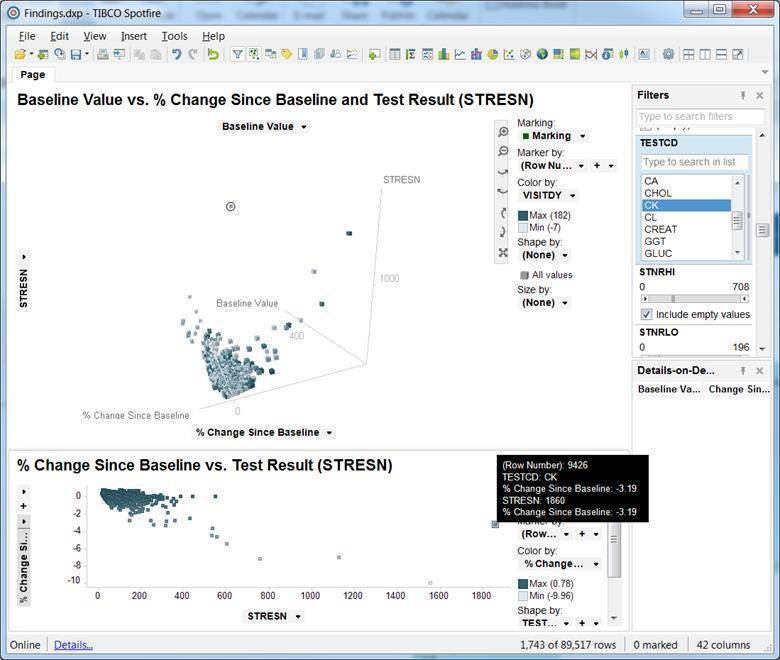
- Use visualization tools like Spotfire to produce interactive and actionable graphs
By actionable, I mean that the user should be able to highlight outliers or specific data points and be able to take an action, such as Mark as Reviewed or Create a Query.
Visualization tools can effectively be used to quickly explore and highlight outliers and questionable data, as shown in the following examples of scatter plots:
Stay tuned for my next post, in which we’ll discuss leveraging value added data for your clinical data review.
In the meantime, if you are interested in learning how my team can help improve your clinical data review process, please send us an email.